| Basic Information | |
| What is this? | The Crab Nebula |
| Where is it in the sky? | In the constellation of Taurus |
| How big is it? | Around 10 light years across |
| How far away is it? | Around 6500 light years away |
| What do the colours represent? | Blue and purple shows visible light seen by the Hubble Space Telescope, showing hot gas. Red and pink shows cool dust as seen by Herschel |
Downloads
See this object in:
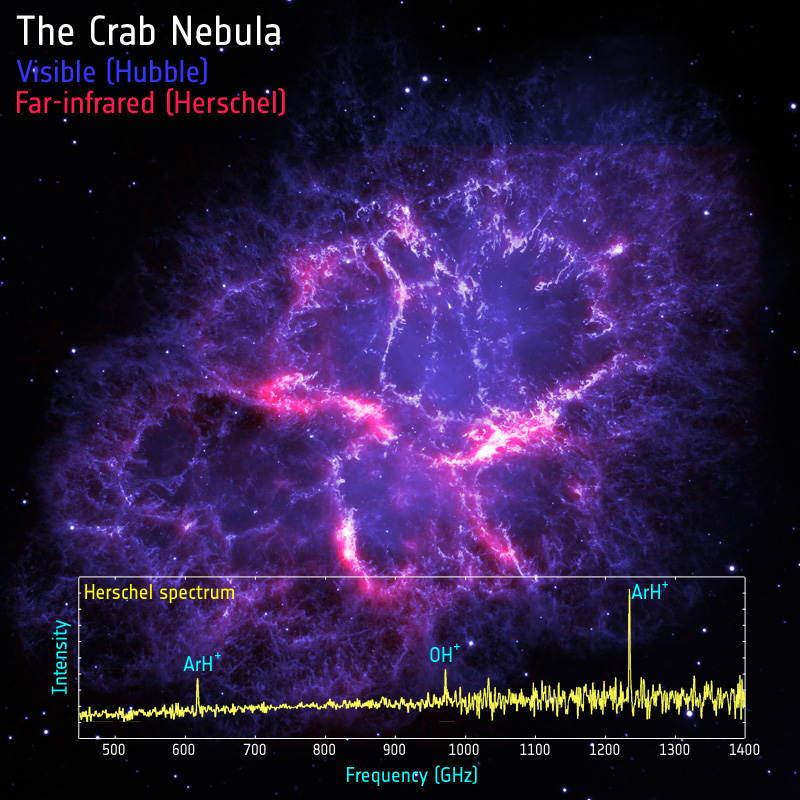
Astronomers using Herschel have made the first discovery in space of a molecule including a noble gas. The molecule, argon hydride, was seen in the Crab Nebula, the remains of a star that exploded 1000 years ago. Such molecules have only been studied in laboratories before, and finding them in space is a big surprise.
The noble gases, which include helium, argon, radon and krypton, do not react easily with other chemical elements, and so are usually found on their own. But they are not completely unreactive, and in the right circumstances can form molecules with other elements. Such chemical compounds have only ever been studied in laboratories on Earth, and have never been seen out in space. The fact that such molecules had not been found led to the assumption that such conditions simply don’t occur out in space.
Enter the Crab Nebula, which is all that remains of a star which exploded 1000 years ago. As the material has expanded outwards it has formed an intricate network of filaments of gas and dust. It is in these filaments that the team first discovered the molecule argon hydride. “At first, the discovery seemed bizarre,” comments Prof Mike Barlow, from University College London, who led the team which made this discovery.
“With hot gas still expanding at high speeds after the explosion, a supernova remnant is a harsh, hostile environment, and one of the places where we least expected to find a noble-gas based molecule,” he adds.
One way of forming noble-gas molecules is to ionise, or energise, the gas, stripping the atoms of their electrons. It then needs to be mixed with a cold gas, such as molecular hydrogen, made of two hydrogen atoms. The problem is that to ionise a noble gas requires high energies and high temperatures, the very conditions that would rip apart the cold gas into its constituent atoms.
It now seems that the Crab Nebula provides exactly the right conditions to form such molecules, in this case ionised argon hydride – formed when ionised argon meets cold molecular hyrdogen gas. The argon was produced in the initial stellar explosion, and then ionised by the resulting intense radiation as shockwaves. These shockwaves led to the formation of the network of filaments, which have also provided the conditions perfect for cold gas. The densest parts create protective cocoons of dust and gas, shielding isolated pockets from the radiation and allowing cold molecular gas to form.
Watch Prof. Mike Barlow talking about the discovery
“We soon realised that even in the Crab Nebula, there are places where the conditions are just right for a noble gas to react and combine with other elements. There, in the transition regions between ionised and molecular gas, argon hydride can form and survive,” explains Barlow.
As the molecules spin in space, they emit light of very specific wavelengths, or colours, called “emission lines”. The precise wavelength is dictated by the composition and structure of the molecule, and so studying these emission lines allows astronomers to study the chemistry of outer space. These measurements of the Crab Nebula were made using Herschel’s SPIRE instrument, the development and operation of which was led by the UK.
In this case, the team did not set out to make the discovery, but instead stumbled upon it almost by accident. “The discovery was truly serendipitous: we were observing the Crab Nebula to study its dust content. But then, on top of the emission from dust, we found two emission lines that had never been seen before,” says co-author Prof Bruce Swinyard, also from University College London.
But these emission lines tell astronomers a whole lot more than just the chemical composition of the molecules, and can even reveal the isotope, or type, of the elements forming the molecule. “And there’s icing on the cake: from a molecule’s emission, we can determine the isotope of the elements that form it – something that we can’t do when we see only ions,” adds Swinyard. The isotope, or type, of argon in the Crab Nebula was found to be argon-36, which is the main type formed from the nuclear reactions that occur during supernova explosions.
Although the Herschel Spacecraft finished observing in April this year, the team are planning observations with other telescopes, to see if they can identify and measure any other isotopes of argon. Those measurements will provide an insight into the details of the supernova which produced the Crab Nebula nearly 1000 years ago.
“What a great detective story”, says Prof Matt Griffin, from Cardiff University, and lead scientist of the team behind the SPIRE instrument. “Here we see the excellent performance of the Herschel-SPIRE spectrometer, the expertise of the instrument team in producing the highest quality data, and the tenacity and vision of the scientists analysing it, all coming together to make an intriguing new discovery”, he concludes.


Comments are closed.